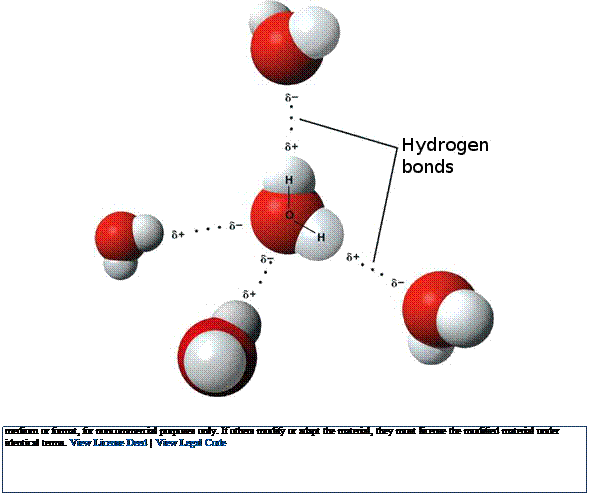
Fig. 2.6 Water molecules. In water and other molecules,
oxygen keeps the
electrons most of the time. This produces positive (+)
and negative (-) regions and
hydrogen bonds. Hydrogen bonds form within and between many body molecules affecting shapes and functions (e.g., DNA, proteins). (For examples, see the following, where hydrogen bonds shown with dotted lines: https://commons.wikimedia.org/wiki/File:DNA_molecular_structure,_showing_individual_nucleotides_and_bonds.jpg; https://commons.wikimedia.org/wiki/File:Alpha_beta_structure_(full).png.) (Images used with permission. See below)
Copyright
2020: Augustine G. DiGiovanna, Ph.D., Salisbury University, Maryland
The materials on this site are licensed under CC BY-NC-SA 4.0 .
![]()
https://www.biologyofhumanaging.com/Figures/CC-BY-NS-SA%20image.jpg
Attribution-NonCommercial-ShareAlike
This license requires that reusers
give credit to the creator. It allows reusers to
distribute, remix, adapt, and build upon the material in any medium or format,
for noncommercial purposes only. If others modify or adapt the material, they
must license the modified material under identical terms.
Previous print editions of the text Human Aging: Biological Perspectives are ©
Copyright 2000, 1994 by The McGraw-Hill Companies, Inc. and 2020 by Augustine
DiGiovanna.
View License Deed | View Legal Code